Submonomer Synthesis of Inverse Polyamidoamine (i-PAMAM) Dendrimer Antibacterials
Check out our latest paper Submonomer synthesis of inverse polyamidoamine (i-PAMAM) dendrimer antibacterials in Helvetica Chimica Acta!
Abstract
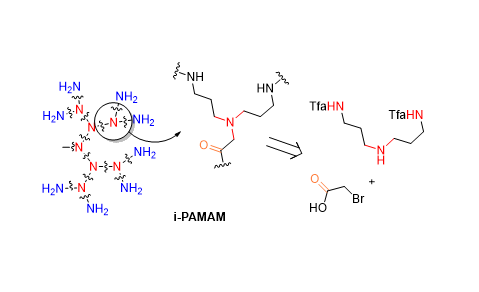
Herein we report that the submonomer method for peptoid synthesis enables access to pure i-PAMAM dendrimers up to 16 amino termini by a divergent solid-phase synthesis using the inexpensive bis(3-trifluoroacetamidopropyl)amine as branching unit. We exemplify this new and efficient approach by a structure-activity relationship study of antibacterial dendrimers obtained by appending the polycationic i-PAMAM dendrimer to a hydrophobic core consisting of either an oligoleucine peptide or an oligo-N-isobutylglycine peptoid. These non-hemolytic dendrimers kill Gram-negative bacteria such as Pseudomonas aeruginosa, Acinetobacter baumannii and Escherichia coli as well as the Gram-positive MRSAby a non-membrane disruptive mechanism involving aggregation of intracellular contents as reported for antimicrobial peptoids.
Author(s): Hippolyte Personne, Xiaoling Hu, Etienne Bonvin, Jérémie Reusser and Jean-Louis Reymond