Synthesis and characterisation of fluorescent substrates for eukaryotic protein N-glycosylation
The paper Synthesis and characterisation of fluorescent substrates for eukaryotic protein N-glycosylation is now available online in Tetrahedron!
Check it out: https://www.sciencedirect.com/science/article/pii/S0040402023001436
Keywords:
Carbohydrates; Fluorescence; Dolichol; Chitobiose; Mannose; Protein glycosylation
Abstract:
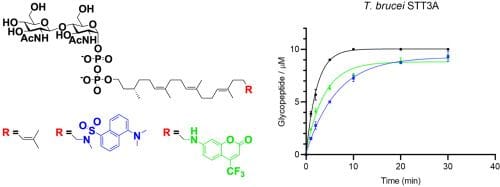
Herein we report the synthesis of two fluorescently labelled analogues of C25 dolichol (Dol25) in which the terminal isoprene unit has been replaced by a dansyl or 7-amino-4-trifluoromethylcoumarin fluorophore, a transformation enabled by the regioselective epoxidation of the terminal olefin via its bromohydrin using the van Tamelen procedure. The lipid alcohols were phosphorylated and glycosylated to obtain lipid-linked chitobiose-α-diphosphates and a lipid-linked mannosyl-β-phosphate. Biochemical assays showed that these labelled substrates are accepted by eukaryotic protein N-glycosylation enzymes with rates comparable to the unlabelled substrates, reconstituting a major part of the pathway up to the lipid-linked dodeca-saccharide Glc1Man9GlcNAc2-PP-Dol25 and its transfer to an acceptor peptide catalysed by eukaryotic oligosaccharyltransferases (OTSs), namely the single-subunit OST STT3A from Trypanosoma brucei and the octameric OST complex from Saccharomyces cerevisiae. The fluorescent labels facilitate handling and purification of the lipid-linked glycosyl donors and acceptors and should facilitate further biochemical studies of protein glycosylation enzymes.
Author(s): Mario M. de Capitani, Ana S. Ramírez, Lorenzo Rossi, J. Andrew, N. Alexander, Sabrina De Lorenzo, Kaspar P. Locher, and Jean-Louis Reymond