Rotavirus Spike Protein VP4 Mediates Viroplasm Assembly by Association to Actin Filaments
Our paper Rotavirus Spike Protein VP4 Mediates Viroplasm Assembly by Association to Actin Filaments is now published in Journal of Virology!
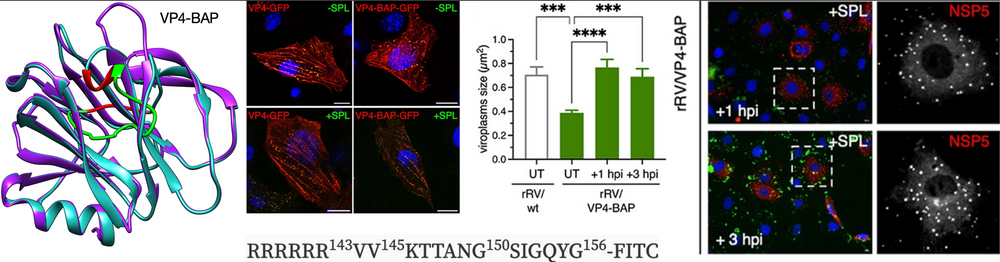
The Institute of Virology, Zurich in collaboration with us performed this research study. With a peptide mimicking loop K145-G150 of VP4 of rotavirus, we were able to link spike protein VP4 to catalysis of viroplasm assembly, which is essential for persistent infection.
A highly contagious virus, rotavirus is a double-stranded RNA virus that causes diarrhoea in children. It is estimated that every child in the world will contract rotavirus at least once before the age of five. It is estimated that around 150,000 children under the age of 5 died from diarrhoea in 2019.
Abstract:
Rotavirus (RV) viroplasms are cytosolic inclusions where both virus genome replication and primary steps of virus progeny assembly take place. A stabilized microtubule cytoskeleton and lipid droplets are required for the viroplasm formation, which involves several virus proteins. The viral spike protein VP4 has not previously been shown to have a direct role in viroplasm formation. However, it is involved with virus-cell attachment, endocytic internalization, and virion morphogenesis. Moreover, VP4 interacts with actin cytoskeleton components, mainly in processes involving virus entrance and egress, and thereby may have an indirect role in viroplasm formation. In this study, we used reverse genetics to construct a recombinant RV, rRV/VP4-BAP, that contains a biotin acceptor peptide (BAP) in the K145-G150 loop of the VP4 lectin domain, permitting live monitoring. The recombinant virus was replication competent but showed a reduced fitness. We demonstrate that rRV/VP4-BAP infection, as opposed to rRV/wt infection, did not lead to a reorganized actin cytoskeleton as viroplasms formed were insensitive to drugs that depolymerize actin and inhibit myosin. Moreover, wild-type (wt) VP4, but not VP4-BAP, appeared to associate with actin filaments. Similarly, VP4 in coexpression with NSP5 and NSP2 induced a significant increase in the number of viroplasm-like structures. Interestingly, a small peptide mimicking loop K145-G150 rescued the phenotype of rRV/VP4-BAP by increasing its ability to form viroplasms and hence improve virus progeny formation. Collectively, these results provide a direct link between VP4 and the actin cytoskeleton to catalyze viroplasm assembly.
Author(s): Janine Vetter, Guido Papa, Michael Seyffert, Kapila Gunasekera, Giuditta De Lorenzo, Mahesa Wiesendanger, Jean-Louis Reymond, Cornel Fraefel, Oscar R. Burrone and Catherine Eichwald