Chemo-enzymatic synthesis of lipid-linked GlcNAc2Man5 oligosaccharides using recombinant Alg1, Alg2 and Alg11 proteins
The paper Chemo-enzymatic synthesis of lipid-linked GlcNAc2Man5 oligosaccharides using recombinant Alg1, Alg2 and Alg11 proteins has been accepted for publication by Glycobiology.
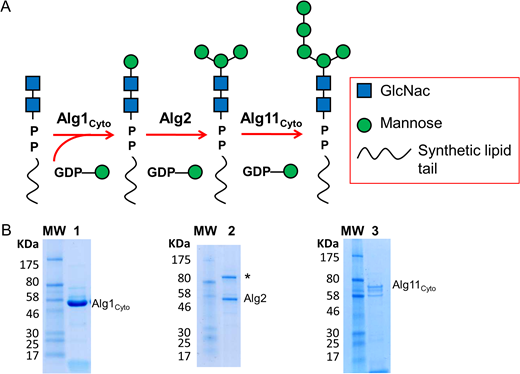
The biosynthesis of eukaryotic lipid-linked oligosaccharides (LLOs) that act as donor substrates in eukaryotic protein N-glycosylation starts on the cytoplasmic side of the endoplasmic reticulum and includes the sequential addition of five mannose units to dolichol-pyrophosphate-GlcNAc2. These reactions are catalyzed by the Alg1, Alg2 and Alg11 gene products and yield Dol-PP-GlcNAc2Man5, an LLO intermediate that is subsequently flipped to the lumen of the endoplasmic reticulum. While the purification of active Alg1 has previously been described, Alg11 and Alg2 have been mostly studied in vivo. We here describe the expression and purification of functional, full length Alg2 protein. Along with the purified soluble domains Alg1 and Alg11, we used Alg2 to chemo-enzymatically generate Dol-PP-GlcNAc2Man5 analogs starting from synthetic LLOs containing a chitobiose moiety coupled to oligoprenyl carriers of distinct lengths (C10, C15, C20 and C25). We found that while the addition of the first mannose unit by Alg1 was successful with all of the LLO molecules, the Alg2-catalyzed reaction was only efficient if the acceptor LLOs contained a sufficiently long lipid tail of four or five isoprenyl units (C20 and C25). Following conversion with Alg11, the resulting C20 or C25 -containing GlcNAc2Man5 LLO analogs were successfully used as donor substrates of purified single-subunit oligosaccharyltransferase STT3A from Trypanosoma brucei. Our results provide a chemo-enzymatic method for the generation of eukaryotic LLO analogs and are the basis of subsequent mechanistic studies of the enigmatic Alg2 reaction mechanism.
Author(s): Ana S Ramírez, Jérémy Boilevin, Chia-Wei Lin, Bee Ha Gan, Daniel Janser, Markus Aebi, Tamis Darbre, Jean-Louis Reymond, Kaspar P Locher