Adaptive and mutational responses to peptide dendrimer antimicrobials in Pseudomonas aeruginosa
The Article Adaptive and mutational responses to peptide dendrimer antimicrobials in Pseudomonas aeruginosa was published in Antimicrobial Agents and Chemotherapy.
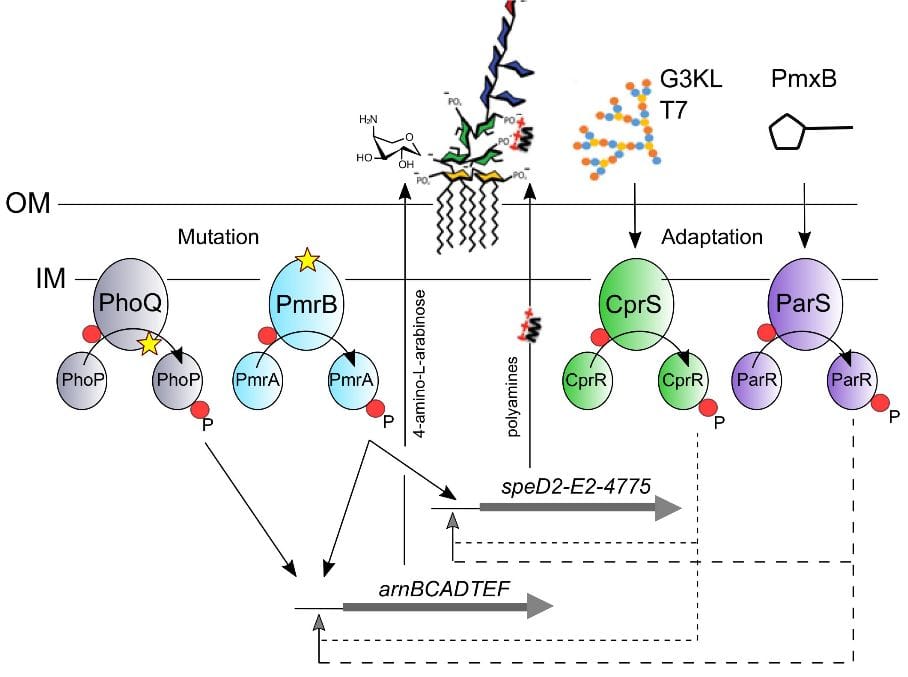
Colistin (polymyxin E) is a last resort antibiotic against multidrug-resistant isolates of Pseudomonas aeruginosa. However, the nephro-toxicity of colistin limits its use, spurring the interest in novel antimicrobial peptides (AMP). Here we show that the synthetic dendrimer AMPs G3KL (MW 4531.38, 15 positive charges, MIC = 8 mg/L) showed faster killing than polymyxin B (Pmx-B) and no detectable resistance selection in P. aeruginosa strain PA14. Spontaneous mutants selected on Pmx-B, harboring loss of function mutations in the PhoQ sensor kinase gene, showed increased Pmx-B MICs and arnB operon expression (4-amino-L-arabinose addition to lipid A), but remained susceptible to dendrimers. Two mutants carrying a missense mutation in the periplasmic loop of the PmrB sensor kinase showed increased MICs for Pmx-B (8-fold) and G3KL (4-fold) but not for the dendrimer T7 (MW 4885.64, 16 positive charges, MIC = 8 mg/L). The pmrB mutants showed increased expression of the arnB as well as of the speD2-speE2-PA4775 operon, located upstream of pmrAB, and involved in polyamine biosynthesis. Exogenous supplementation with the polyamines spermine and norspermine increased G3KL and T7 MICs in a phoQ mutant background but not in the PA14 wild-type. This suggests that both addition of 4-amino-L-arabinose and secretion of polyamines are required to reduce susceptibility to dendrimers, probably neutralizing the negative charges present on the lipid A and the KDO sugars of the LPS, respectively. We further show by transcriptome analysis that the dendrimers G3KL and T7 induce adaptive responses through the CprRS two-component system in PA14.
Author(s): Fatma Ben Jeddou, Léna Falconnet, Alexandre Luscher, Thissa Siriwardena, Jean-Louis Reymond, Christian van Delden, Thilo Köhler